Peptide and protein microarray immunoprofiling for complex vaccines and biologics
We help vaccine and biologic teams generate clearer, more actionable insights into immune responses. Using advanced peptide and protein microarrays, we capture thousands of antibody binding patterns from small volumes of serum samples.Our integrated machine learning and AI approaches translate these complex immunoprofiling data into practical predictions, such as early immunogenicity signals, response durability and antigen prioritisation.This helps de‑risk your pipeline and speed better decisions, especially for challenging multi‑component or bacterial vaccines and complex biologics.
Subscribe to ImmunoSig on Substack for vaccine insights
ImmunoSig Ltd. is a company incorporated in England and Wales.
Company No. 16642090, VAT No. GB 510 1556 40.
3rd Floor 86-90 Paul Street, London, EC2A 4NE
© 2026 All rights reserved. ImmunoSig Ltd. Privacy Notice
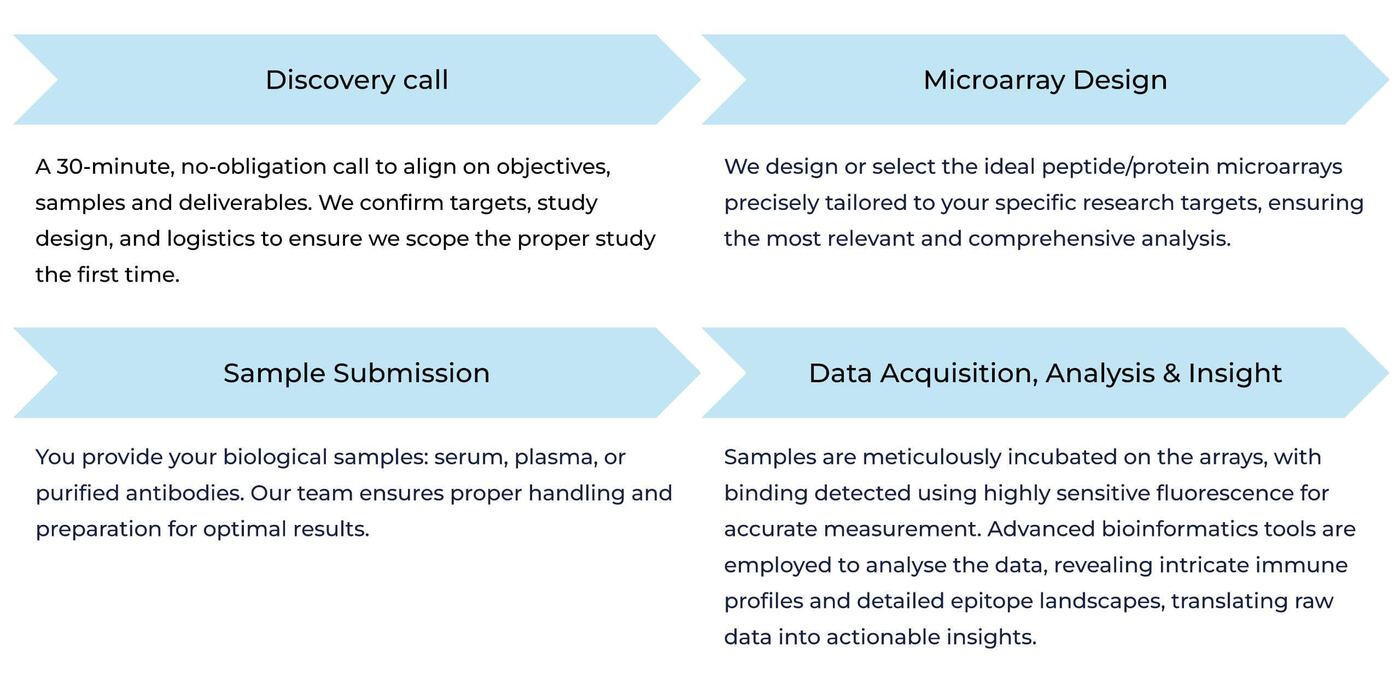
Our biologic & vaccine immunoprofiling platformHow It Works: From Sample to Insight
How It Works: Biologic formulation package
How It Works: Immunoprofiling package
About ImmunoSig Ltd.
ImmunoSig Ltd was founded by experienced academic and industry scientists with deep expertise in vaccine immunology, protein microarrays and biologics development. Our team has led projects with global vaccine institutes, industry partners and major funders, and we bring that experience directly into our CRO work.

Prof. Jeremy Derrick
Co-Founder
Jeremy obtained his first degree and PhD from the University of Cambridge. He was appointed a Lecturer at UMIST in Manchester in 1993 and was a Lister Institute Fellow from 1996 to 2001. He joined the University of Manchester in 2003, when the two universities merged. He has worked extensively on protein structure and function, publishing more than 130 peer-reviewed papers. He has served on multiple grant panels, and is continually requested to act as a reviewer for major scientific journals. More recently, he has applied his extensive commercial experience to projects in immunoprofiling, vaccine development, and biologic formulation

Dr Fidel Ramirez Bencomo
Co-founder
Fidel graduated in Biochemistry and Molecular Biology from the University of Havana and earned a PhD in Microbiology from the University of Manchester. With over 10 years’ experience, he specialises in monoclonal antibody production, diagnostic assay development, and protein microarrays. Fidel’s work at the Finlay Vaccine Institute (Cuba) and in the University of Manchester focuses on immunoprofiling, epitope mapping, and vaccine development. He has led projects supported by GSK and the Bill and Melinda Gates Foundation, and has presented at major conferences and published numerous peer-reviewed papers.
Discuss your immunoprofiling needs
Ready to plan a vaccine immunoprofiling or biologics immunogenicity study? Share a few details about your project, and we’ll suggest how peptide and protein microarrays could support your next decision point.
Thank you
Thank you for your message. We will reply to you within 2-3 business days.
Thank you
Thank you for requesting our launch webinar. The link will be landing in your inbox soon. Looking forward to your feedback.
Privacy notice
ImmunoSig Ltd is committed to protecting your privacy. We collect only the information necessary to provide our services, communicate with clients, and maintain regulatory compliance. Any personal data shared with us is processed securely and never sold or disclosed to third parties, except where required by law or for the delivery of essential services.You can review the full details of our data practices, retention policies, and your rights under UK GDPR in our Privacy Policy document below.
Frequently Asked Questions (FAQs)
Launch webinar
In our launch webinar we showed how our peptide and protein microarray platform delivers detailed immunoprofiles for vaccines and biologics. Request the recording to see example study designs and data outputs
Our Immunoprofiling Services
We provide specialised immunoprofiling services built around high‑density peptide and protein microarrays. Our platform is designed for vaccine and biologics teams who need more than standard ELISAs or a single titer readout. By capturing detailed epitope‑level antibody responses, we help you understand breadth, specificity and cross‑reactivity in a way that directly supports decision‑making in development.
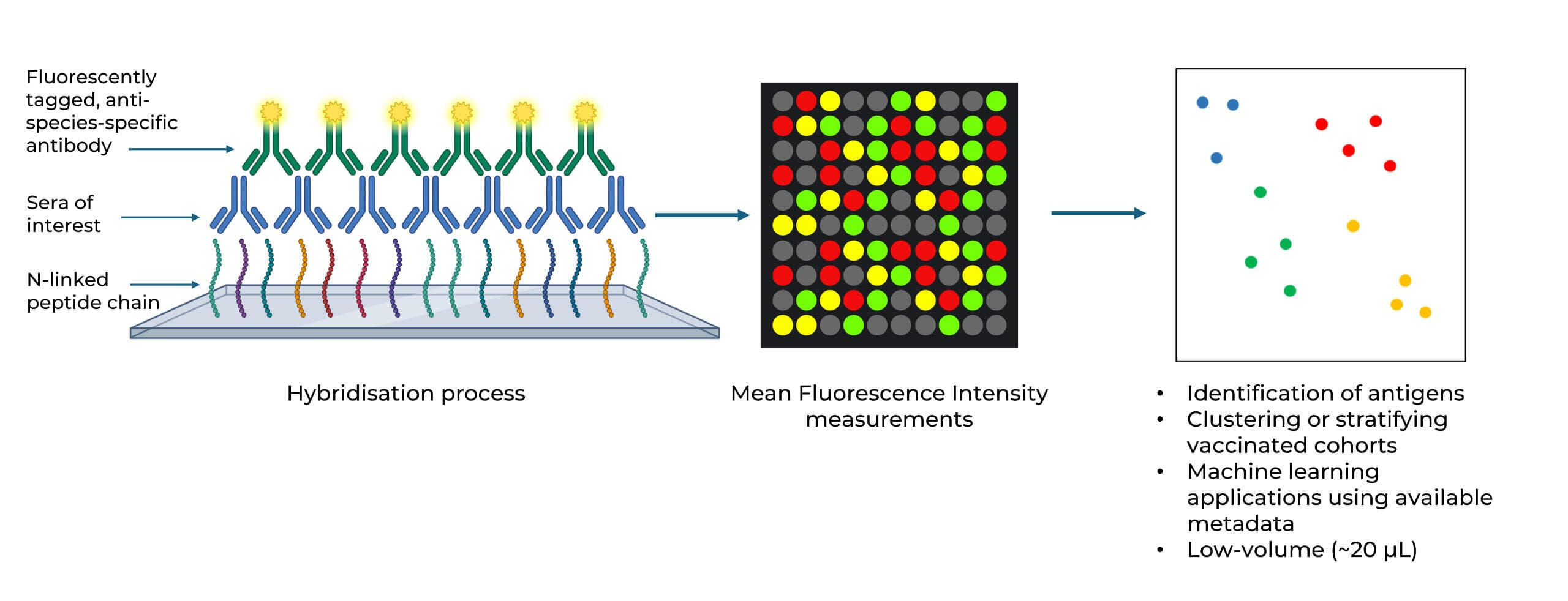
Vaccine Immunoprofiling
We characterise humoral immune responses to complex vaccines using high-density peptide and protein microarrays. Our platform maps antibody epitope specificity, cross-reactivity, and response breadth across viral and bacterial antigens, including OMV, mRNA, viral vector, and multi-component vaccine platforms.
Peer-reviewed and published in Nature Communications (2024), Vaccine (2025), PLoS Pathogens (2024) and Journal of Infectious Diseases (2025).
Biologics Immunogenicity Profiling
We use peptide and protein microarrays to identify immunogenic epitopes, characterise anti-drug antibody responses, and assess comparability following manufacturing or formulation changes. This supports early immunogenicity risk assessment for monoclonal antibodies, bispecific antibodies, and other complex biologics.
Applying established peptide and protein microarray methodology to immunogenicity assessment for therapeutic antibodies and complex biologics.
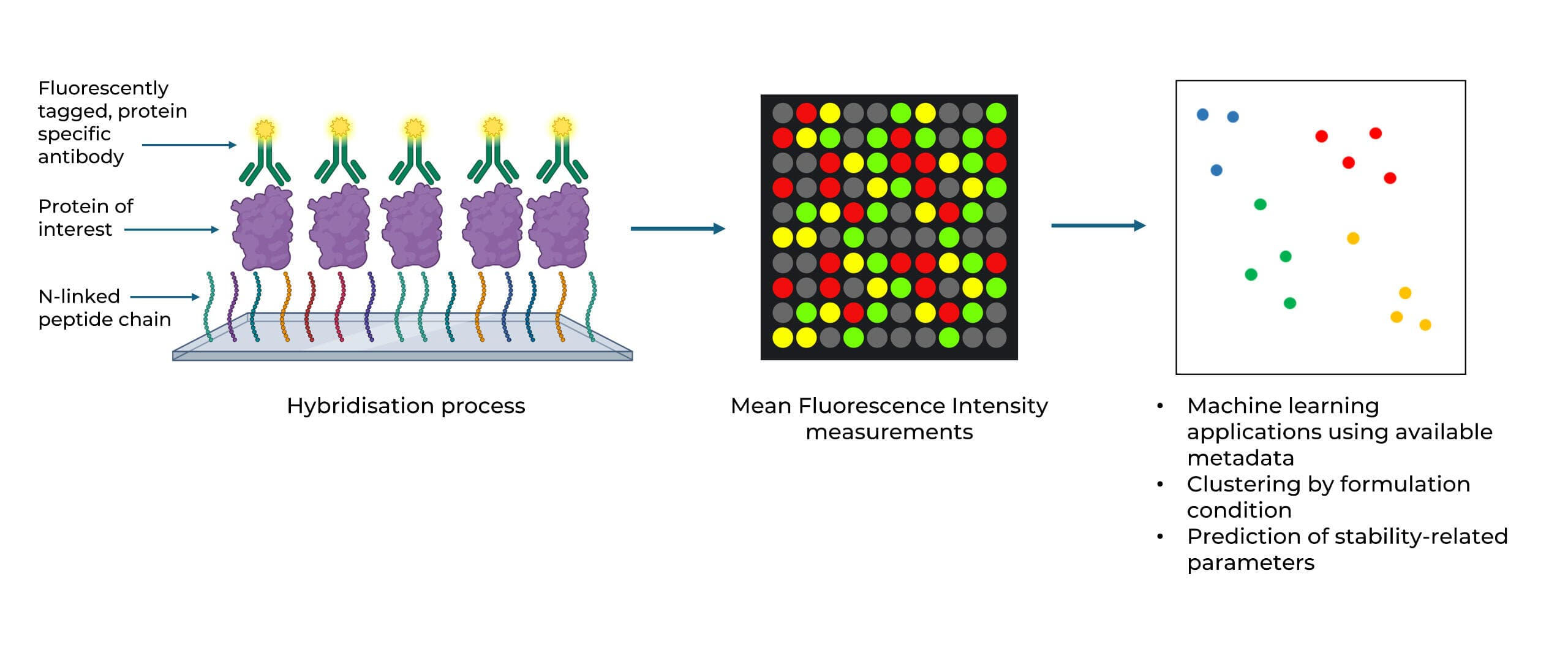
Biologic Formulation Profiling
We use peptide microarrays to quickly profile how therapeutic antibodies and other biologics perform under various formulation conditions, including buffer type, pH, salt concentration, and excipients. Machine learning analysis of binding fingerprints identifies the best formulation options and predicts stability indicators such as aggregation temperature and self-association, using only microgram quantities of material.
Peer-reviewed and published in Molecular Pharmaceutics (2021).
Immunosignature Discovery
Using high-density peptide and protein microarrays combined with machine learning, we develop immunosignatures, which are patterns of antibody binding that correlate with protection, efficacy, or immunogenicity risk. This systems-level approach offers insights into vaccine and biologic responses that single-antigen assays cannot reveal.
Applied in the characterisation of antigenic diversity and epitope-level immune responses across pathogen populations (mBio, 2025).
Our Platform in Peer-Reviewed Science
ImmunoSig's founders have developed and validated this platform through peer-reviewed studies published in leading journals in collaboration with GSK, the University of Oxford, the FDA, the NIH, and the Wellcome Trust.
Nature Communications · 2024
Profiling IgG and IgA antibody responses during vaccination and natural infection with Neisseria gonorrhoeae using antigen microarrays. Wellcome Trust-funded clinical study in Kenya in collaboration with the University of Oxford and KEMRI.
Stejskal, Thistlethwaite, Ramirez-Bencomo et al. → View on PubMed
PLoS Pathogens · 2024
Tackling immunosuppression by Neisseria gonorrhoeae to improve vaccine design, using microarray profiling to characterise how engineered OMV compositions reshape antibody responses and antigen recognition breadth.
Jones, Ramirez-Bencomo et al. → View on PubMed
Vaccine · 2025
Identification of immunogenic outer membrane vesicle antigens in the licensed meningococcal vaccine Bexsero® using protein microarray profiling of sera from a GSK phase II clinical trial.
Ramirez-Bencomo et al. → View on PubMed
mBio · 2025
Investigating genetic, antigenic and structural diversity of the Neisseria gonorrhoeae outer membrane across 22,000+ isolates, using microarray-based immune response mapping to identify conserved and variable epitopes for vaccine design.
Harrison, Ramirez-Bencomo et al. → View on PubMed
Journal of Infectious Diseases · 2025
Evaluation of immunisation route in induction of vaccine-mediated anti-gonococcal immune responses in a murine model of ascending infection — characterising IgG, IgA and opsonophagocytic activity using antigen microarrays.
Matthias, Ramirez-Bencomo et al. → View on PubMed
Molecular Pharmaceutics · 2021
Use of peptide microarrays for fast and informative profiling of therapeutic antibody formulation conditions — demonstrating that binding fingerprints combined with machine learning can predict aggregation temperature, self-association and stability indicators from microgram quantities of material.
Austerberry, Derrick et al. → View on PubMed
